Beatrice Cobucci-Ponzano
Role: Senior Researcher
Section: Researchers and Technologists
Division: Naples
Tel: (39) 081-6132564
E-mail: beatrice.cobucciponzano[at]cnr.it
URL: http://ibbr.cnr.it

Address
Dr. Beatrice Cobucci-Ponzano
Senior Researcher
Via P. Castellino 111, 80131 Naples ITALY
tel. +39 081 6132564 (direct); +39 081 6132563 (lab)
e-mail: beatrice.cobucciponzano@ibbr.cnr.it
ORCID iD https://orcid.org/0000-0002-8211-2297
National Coordinator fo the Research Infrastructure of biotechnology IBISBA-IT
Member of the steering committee of the Italian Society of Astrobiology
Research Interests
Our group aims to study the structure/function relationship of proteins and enzymes and the mechanistic aspects related to hydrolysis and enzymatic synthesis of glycosides. In particular, we are interested in enzymes that modify carbohydrates (CAZymes) and our studies are also aimed at metagenomic analyzes of extreme environments for the identification of new hyperstable glycosidases for second and third generation biorefineries and for green chemistry.
In the last ten years, the study of the stabilization of glycosidases has included the human enzymes whose deficiency causes lysosomal storage diseases and methods to recover their activity for therapeutic purposes.
In addition, studying the gene of an archaeal alpha-fucosidase interrupted by a frameshifting -1, we demonstrated that the gene is translated by following a mechanism of regulation of gene expression named programmed -1 frameshifting. From then, the studies on extremophilic Archaea and on the mechanism of regulation of translation of interrupted genes in relation with the limits of life and evolution in extreme environments continue thanks to several funded projects on astrobiology.
Our lab belongs to the European Research Infrastructure IBISBA-EU.
Our lab belongs to the Società italiana di astrobiologia (SIA) and is part of the national network of astrobiology lab (IAI-ASTROBIOLab).
Communication and dissemination activities
Intervista RAI Scuola - Newton - La vita oltre la Terra St 2022 Ep 17
Intervista RAI Scuola - Storie della Scienza - Origine della Vita St 2022 Ep 15
Intervista Radio 24 - Smart City - Estremofili per la sostenibilità
Intervista Radio 24 - Smart City - Cianobatteri su Marte: con un "aiutino" pot...
Extreme tour, Dai Greci, ai fasti del Grand Tour alla ricerca della vita su Ma...
CAZymes Discovery

The enzymes that modify carbohydrates (or cazymes, acronym for carbohydrate active enzymes) represent a very interesting class of enzymatic activities, with multiple applications, which are classified in the Carbohydrate Active enZYmes database (CAZY, www.cazy.org) in families that group enzymes with reaction mechanism, catalytic residues and common structure, but different specificity. The enzymatic characterization of unknown sequences and the identification of new enzymatic activities associated with known proteins, gives CAZy the possibility to improve the classification, create new families and better understand their characteristics.
In addition, the rapid progress of omic technologies, particularly of (meta) genomics, has produced an exponential increase in sequence data. However, many of the annotated sequences belong to enzymes and proteins that are not yet characterized or that, due to annotation errors, are cataloged incorrectly. It is therefore imperative for protein biochemists and enzymologists to do their utmost to characterize as many genome (meta) sequences as possible in an attempt to assign them a function and therefore identify innovative enzymes with important applications in second and third generation biorefineries and for green chemistry.
Glycosynthases

CAZymes represent an interesting class of enzymatic activities for the synthesis of oligosaccharides. In particular, glycosyl hydrolases (GH), while hydrolyzing sugars, under suitable conditions, can carry out transglycosylation reactions where a ’donor’ is transferred to a sugar / alcohol instead of water. Under these conditions, however, yields rarely exceed 50%, as the products become substrates of the enzyme. We contributed in carrying on the studies started by Prof. Marco Moracci on the modification of glycosidases in glycosyntases makingfor the first time innovative alpha-glycosyntases (alpha-fucosyntasesand alpha-galactosyntases), enzymes that had hitherto proved refractory to conversion to glycosyntases.
Pharmacological Chaperones
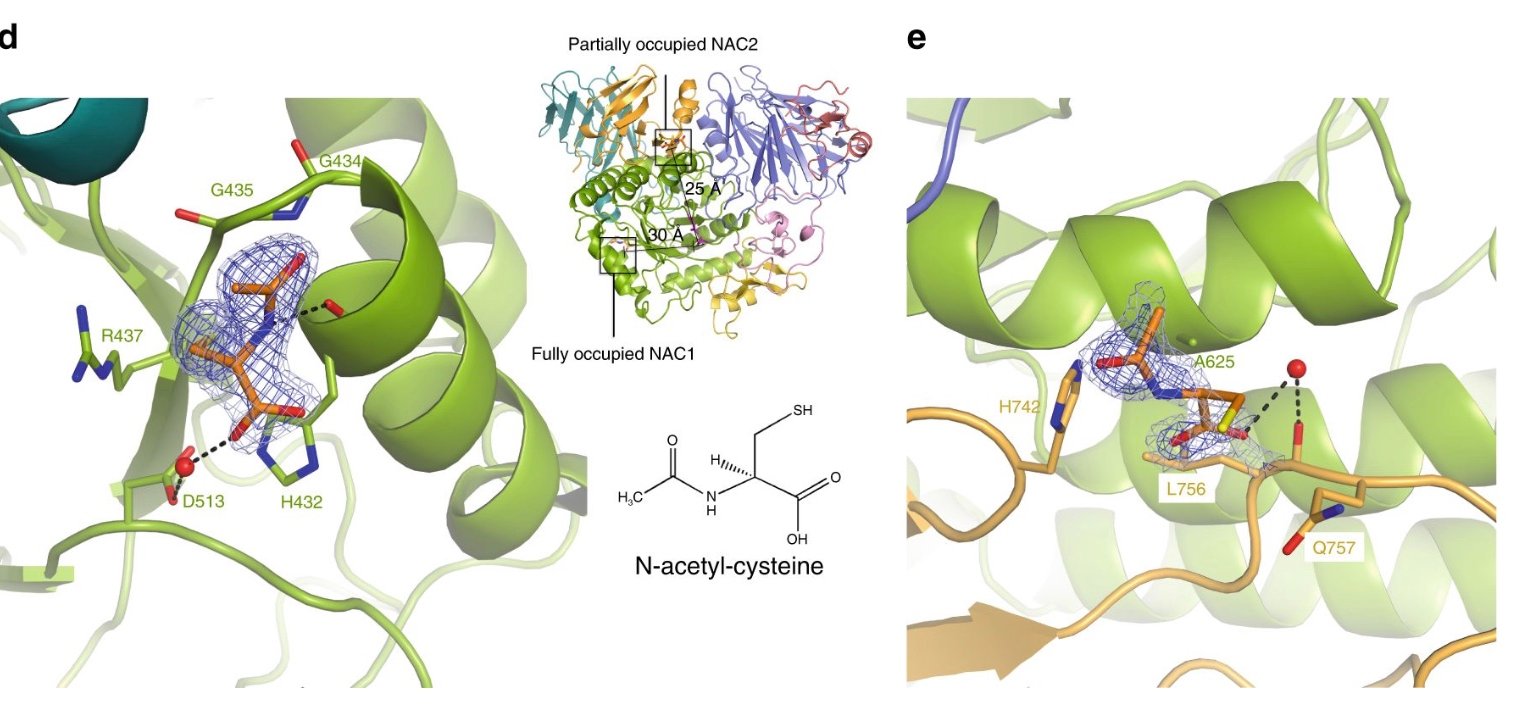
At the beginning of my scientific career I was involved in the study of the molecular mechanisms of stabilization of enzymes from hypertemophilic microorganisms (which only grow at T> 80 ° C), especially from Archaea. More recently, the study of the stabilization of glycosidases has turned to human enzymes whose deficiency causes lysosomal storage diseases and methods to recover their activity for therapeutic purposes. In particular, in collaboration, she is working on human acid alpha-glucosidase (GAA), whose deficiency determines type II glucogenosis (Pompe disease). One of the most promising therapeutic approaches for the treatment of this and other, orphan disease is the use of small molecules which, by binding to the misfolded alpha-glucosidase, function as pharmacological chaperones (PC), presumably stabilizing its structure and allowing its vehiculation to the lysosome to hydrolyze the glycogen. In collaboration, we demonstrated that N-acetylcysteine is a new PC. These studies led to the filing of two patents and to the resolution, for the first time, of the 3D structure of the human GAA.
Translational Recoding
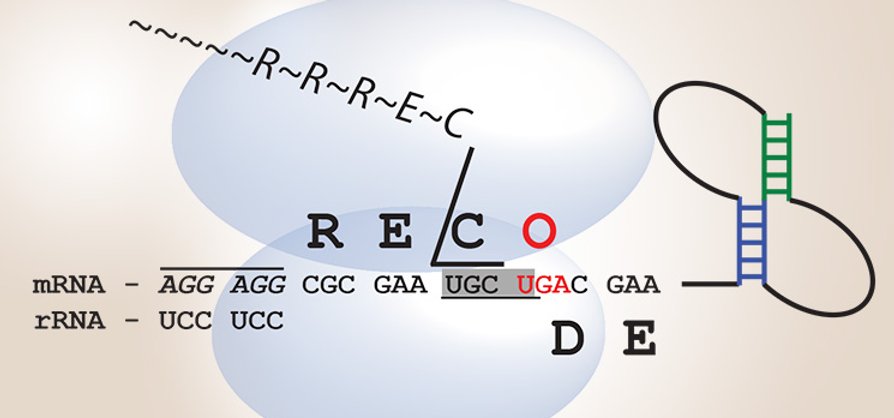
Some years ago, studying the gene of an archaeal alpha-fucosidase interrupted by a -1 frameshifting, we showed that the expression of the gene was corrected at translational level both inE. coli and in S. solfataricuswith a programmed frameshifting mechanism. This process, known in Bacteria and Eukaryotes (especially viruses), is part of the so-called translational recoding, a set of gene expression regulation phenomena in which the ribosome deviates, in finely regulated way, by the "universal" rules of translation. Recoding, in addition to programmed frameshifting +/- 1, includes ribosome hopping, and the assignment of codons to the amino acids 21 ° and 22 ° selenocysteine and pyrrolysine. The study of the alpha-fucosidase has allowed to identify this phenomenon for the first time in the Archaea, and subsequent proteomic studies have shown its wide diffusion in this Domain of life and therefore in all living organisms.
Following the recent progress, our understanding of the flexibility of the genetic code is greatly improved in the last decade. Indeed, it has been widely proven that many genes disrupted, usually considered pseudogenes, are actually functional genes whose expression is finely regulated by translational recoding. Increasing evidences suggest that the genetic code flexibility is a trait selected during evolution and that this gene organization could provide an evolutionary advantage in extreme conditions, expanding the limits of life. With this in mind, the study of the genes interrupted in the model system Saccharolobus under stress and simulated space conditions is of great interest for the study of the evolution of the genetic code and the limits of life from an astrobiological point of view. This work is actually funded by the Italian Space Agency.
Funded Projects
2022 - ITINERIS - Italian Integrated Environmental Research Infrastructures System - . PIANO NAZIONALE DI RIPRESA E RESILIENZA (PNRR) MISSIONE 4, COMPONENTE 2, INVESTIMENTO 3.1 “Fondo per la realizzazione di un sistema integrato di infrastrutture di ricerca e innovazione”
2022 - Bioindustry 4.0. HORIZON-INFRA-2022-TECH-01 I Grant agreement ID: 101094287. DOI 10.3030/101094287
2019-2023 - PREP-IBISBA Grant agreement ID: 871118 - INFRADEV-02-2019-2020 - Preparatory Phase of new ESFRI projects and early phase support to ESFRI high strategic potential areas. H2020
2019-2022 - OPPS LIFE IN SPACE - ORIGIN, PRESENCE, PERSISTENCE OF LIFE IN SPACE, FROM MOLECULES TO EXTREMOPHILES - Italian Space Agency
2017- 2021 IBISBA 1.0 Grant agreement ID: 730976 INFRAIA-02-2017 - Integrating Activities for Starting Communities - H2020
10 Selected Publications
2019 Curci N, Strazzulli A, De Lise F, Iacono R, Maurelli L, Dal Piaz F, Cobucci-Ponzano B*, Moracci M. Identification of a novel esterase from the thermophilic bacterium Geobacillus thermodenitrificans NG80-2. Extremophiles. 23:407-419. doi: 10.1007/s00792-019-01093-9.
2018 Iacono R, Strazzulli A, Maurelli L, Curci N, Casillo A, Corsaro MM, Moracci M, Cobucci-Ponzano B.* A GlcNAc de-N-acetylase from the hyperthermophilic archaeon Sulfolobus solfataricus. Appl Environ Microbiol. In press doi: 10.1128/AEM.01879-18
2017 Roig-Zamboni V, Cobucci-Ponzano B, Iacono R, Ferrara MC, Germany S, Bourne Y, Parenti G, Moracci M, Sulzenbacher G. Structure of human lysosomal acid alpha-glucosidase - a guide for the treatment of Pompe disease. Nat Commun. 8, 1111. doi: 10.1038/s41467-017-01263-3.
2017 Strazzulli A, Fusco S, Cobucci-Ponzano B, Moracci M, Contursi P. Metagenomics of microbial and viral life in terrestrial geothermal environments. Rev Environ Sci Biotechnol 16 425-454 doi: 10.1007/s11157-017-9435-0
2015 Cobucci-Ponzano B., Strazzulli A., Iacono R., Masturzo G., Giglio R., Rossi M., Moracci M. Novel thermophilic hemicellulases for the conversion of lignocellulose for second generation biorefineries. Enzyme Microbial Technology 78, 63-73. doi:10.1016/j.enzmictec.2015.06.014
2012 Porto C, Ferrara MC, Meli M, Acampora E, Avolio V, Rosa M, Cobucci-Ponzano B, Colombo G, Moracci M, Andria G, Parenti G. Pharmacological Enhancement of α-Glucosidase by the Allosteric Chaperone N-acetylcysteine. Molecular Therapy. 20, 2201-11. doi: 10.1038/mt.2012.152.
2012 Cobucci-Ponzano B, Moracci M. (2012) Glycosynthases as tools for the production of glycan analogs of natural products. Natural Product Reports, 29, 697-709. doi: 10.1039/c2np20032e
2010 Cobucci-Ponzano B, Aurilia V, Riccio G, Henrissat B, Coutinho PM, Strazzulli A, Padula A, Corsaro MM, Pieretti G, Pocsfalvi G, Fiume I, Cannio R, Rossi M, Moracci M. A new archaeal beta-glycosidase from Sulfolobus solfataricus: seeding a novel retaining beta-glycan-specific glycoside hydrolase family along with the human non-lysosomal glucosylceramidase GBA2. Journal Biological Chemistry, 285, 20691-20703. doi: 10.1074/jbc.M109.086470
2009 Cobucci-Ponzano B, Conte F, Bedini E, Corsaro MM, Parrilli M, Sulzenbacher G, Lipski A, Dal Piaz F, Lepore L, Rossi M, Moracci M. beta-Glycosyl azides as substrates for alpha-glycosynthases: preparation of efficient alpha-L-fucosynthases. Chemistry and Biolology. 16, 1097-1108. doi: 10.1016/j.chembiol.2009.09.013
2006 Cobucci-Ponzano B., Conte F., Benelli D., Londei P., Flagiello A., Monti M., Pucci P. Rossi M. and Moracci. M. “The gene of an archaeal α-L-fucosidase is expressed by translational frameshifting” Nucleic Acids Research, 34, 4258-4268. doi: 10.1093/nar/gkl574
Selected Publications
(full list available at CNR People)
Carnitine is a pharmacological allosteric chaperone of the human lysosomal α -glucosidase
Iacono R, Minopoli N, Ferrara MC, Tarallo A, Damiano C, Porto C, Strollo S, Roig-Zamboni V, Peluso G, Sulzenbacher G, Cobucci-Ponzano B, Parenti G, Moracci M
Year: 2021
Spatial metagenomics of three geothermal sites in pisciarelli hot spring focusing on the biochemical resources of the microbial consortia
Iacono R, Cobucci-Ponzano B, De Lise F, Curci N, Maurelli L, Moracci M, Strazzulli A
Year: 2020
The Italian National Project of Astrobiology Life in Space Origin, Presence, Persistence of Life in Space, from Molecules to Extremophiles
Onofri S, Balucani N, Barone V, Benedetti P, Billi D, Balbi A, Brucato JR, Cobucci-Ponzano B, Costanzo G, Rocca NL, Moracci M, Saladino R, Vladilo G, Albertini N, Battistuzzi M, Bloino J, Botta L, Casavecchia P, Cassaro A, Claudi R, Cocola L, Coduti A, Di Donato P, Di Mauro E, Dore L, Falcinelli S, Fulle M, Ivanovski S, Lombardi A, Mancini G, Maris M, Maurelli L, Murante G, Negri R, Pacelli C, Pagano I, Piccinino D, Poletto L, Prantera G, Puzzarini C, Rampino S, Ripa C, Rosi M, Sanna M, Selbmann L, Silva L, Skouteris D, Strazzulli A, Tasinato N, Timperio AM, Tozzi A, Tozzi GP, Trainotti L, Ugliengo P, Vaccaro L, Zucconi L
Year: 2020
ENEA, a peach and apricot IgE-binding protein cross-reacting with the latex major allergen Hev b 5
Giangrieco I, Ricciardi T, Alessandri C, Farina L, Crescenzo R, Tuppo L, Ciancamerla M, Rafaiani C, Bernardi ML, Digilio AF, Cobucci-Ponzano B, Tamburrini M, Mari A, Ciardiello MA
Year: 2019
GlcNAc De-N-Acetylase from the Hyperthermophilic Archaeon Sulfolobus solfataricus
Iacono R, Strazzulli A, Maurelli L, Curci N, Casillo A, Corsaro MM, Moracci M, Cobucci-Ponzano B
Year: 2019
Ten years of CAZypedia: a living encyclopedia of carbohydrate-active enzymes
Abbott W, Alber O, Bayer E, Berrin J-G, Boraston A, Brumer H, Brzezinski R, Clarke A, Cobucci-ponzano B, Cockburn D, Coutinho P, Czjzek M, Dassa B, Davies GJ, Eijsink V, Eklof J, Felice A, Ficko-blean E, Pincher G, Fontaine T, Fujimoto Z, Fujita K, Fushinobu S, Gilbert H, Gloster T, Goddard-borger E, Greig I, Hehemann J-H, Hemsworth G, Henrissat B, Hidaka M, Hurtado-guerrero R, Igarashi K, Ishida T, Janecek S, Jongkees S, Juge N, Kaneko S, Katayama T, Kitaoka M, Konno N, Kracher D, Kulminskay A, Van bueren AL, Larsen S, Lee J, Linder M, Loleggio L, Ludwig R, Luis A, Maksimainen M, Mark B, Mclean R, Michel G, Montanier C, Moracci M, Mori H, Nakai H, Nerinckx W, Ohnuma T, Pickersgill R, Piens K, Pons T, Rebuffet E, Reilly P, Remaud-simeon M, Rempel B, Robinson K, Rose D, Rouvinen J, Saburi W, Sakamoto Y, Sandgren M, Shaikh F, Shoham Y, St john F, Stahlberg J, Suits M, Sulzenbacher G, Sumida T, Suzuki R, Svensson B, Taira T, Taylor E, Tonozuka T, Urbanowicz B, Vaaje-kolstad G, Van den ende W, Varrot A, Versluys M, Vincent F, Vocadlo D, Wakarchuk W, Wennekes T, Williams R, Williams S, Wilson D, Withers S, Yaoi K, Yip V, Zhang R
Year: 2018
Formamide-based prebiotic chemistry in the Phlegrean Fields
Botta L, Saladino R, Bizzarri BM, Cobucci-Ponzano B, Iacono R, Avino R, Caliro S, Carandente A, Lorenzini F, Tortora A, Di Mauro E, Moracci M
Year: 2017
N-Butyl-l-deoxynojirimycin (l-NBDNJ): synthesis of an allosteric enhancer of α-glucosidase activity for the treatment of pompe disease
D’alonzo D, De Fenza M, Porto C, Iacono R, Huebecker M, Cobucci-Ponzano B, Priestman DA, Platt F, Parenti G, Moracci M, Palumbo G, Guaragna A
Year: 2017
Conversion of xylan by recyclable spores of Bacillus subtilis displaying thermophilic enzymes
Mattossovich R, Iacono R, Cangiano G, Cobucci-Ponzano B, Isticato R, Moracci M, Ricca E
Year: 2017
Structure of human lysosomal acid α-glucosidase - A guide for the treatment of Pompe disease
Roig-Zamboni V, Cobucci-Ponzano B, Iacono R, Ferrara MC, Germany S, Bourne Y, Parenti G, Moracci M, Sulzenbacher G
Year: 2017
Introducing transgalactosylation activity into a family 42 β-galactosidase
Strazzulli A, Cobucci-Ponzano B, Carillo S, Bedini E, Corsaro MM, Pocsfalvi G, Withers SG, Rossi M, Moracci M
Year: 2017
Metagenomics of microbial and viral life in terrestrial geothermal environments
Strazzulli A, Fusco S, Cobucci-Ponzano B, Moracci M, Contursi P
Year: 2017
Metagenomics of hyperthermophilic environments: biodiversity and biotechnology
Strazzulli A, Iacono R, Giglio R, Moracci M, Cobucci-Ponzano B
Year: 2017
The α-Thioglycoligase derived from a GH89 α- N -Acetylglucosaminidase Synthesises α- N -Acetylglucosamine-based glycosides of biomedical interest
Tshililo NO, Strazzulli A, Cobucci-Ponzano B, Maurelli L, Iacono R, Bedini E, Corsaro MM, Strauss E, Moracci M
Year: 2017
The α-Thioglycoligase derived from a GH89 α-N-Acetylglucosaminidase synthesises α-N-Acetylglucosamine-based glycosides of biomedical interest
Tshililo NO, Strazzulli A, Cobucci-Ponzano B, Maurelli L, Iacono R, Bedini E, Corsaro MM, Strauss E, Moracci M
Year: 2017
Human a-L-fucosidase-1 attenuates the invasive properties of thyroid cancer
Vecchio G, Parascandolo A, Allocca C, Ugolini C, Basolo F, Moracci M, Strazzulli A, Cobucci-Ponzano B, Laukkanen MO, Castellone MD, Tsuchida N
Year: 2017
High-level expression of thermostable cellulolytic enzymes in tobacco transplastomic plants and their use in hydrolysis of an industrially pretreated Arundo donax L. biomass
Castiglia D, Sannino L, Marcolongo L, Ionata E, Tamburino R, De Stradis A, Cobucci-Ponzano B, Moracci M, La Cara F, Scotti N
Year: 2016
(Hyper)thermophilic biocatalysts for second generation biorefineries
Iacono R, Cobucci-Ponzano B, Strazzulli A, Giglio R, Maurelli L, Moracci M
Year: 2016
Novel thermophilic hemicellulases for the conversion of lignocellulose for second generation biorefineries
Cobucci-Ponzano B, Strazzulli A, Iacono R, Masturzo G, Giglio R, Rossi M, Moracci M
Year: 2015
Metagenomics of hyperthermophilic environments: biodiversity and biotechnology
Strazzulli A, Iacono R, Giglio R, Moracci M, Cobucci-Ponzano B
Year: 2015
Glycosidases and Glycosynthases
Cobucci-Ponzano B, Moracci M
Year: 2014
RNA editing and modifications of RNAs might have favoured the evolution of the triplet genetic code from an ennuplet code
Di Giulio M, Moracci M, Cobucci-Ponzano B
Year: 2014
The identification and molecular characterization of the first archaeal bifunctional exo-beta-glucosidase/N-acetyl-beta-glucosaminidase demonstrate that family GH116 is made of three functionally distinct subfamilies
Ferrara MC, Cobucci-Ponzano B, Carpentieri A, Henrissat B, Rossi M, Amoresano A, Moracci M
Year: 2014
Glycosynthases as tools for the production of glycan analogs of natural products
Cobucci-Ponzano B, Moracci M
Year: 2012
Thermophilic glycosynthases for oligosaccharides synthesis
Cobucci-Ponzano B, Perugino G, Strazzulli A, Rossi M, Moracci M
Year: 2012
Translational recoding in archaea
Cobucci-Ponzano B, Rossi M, Moracci M
Year: 2012
Pharmacological Enhancement of α-Glucosidase by the Allosteric Chaperone N-acetylcysteine
Porto C, Ferrara MC, Meli M, Acampora E, Avolio V, Rosa M, Cobucci-Ponzano B, Colombo G, Moracci M, Andria G, Parenti G
Year: 2012
Glycosynthases in Biocatalysis
Cobucci-Ponzano B, Strazzulli A, Rossi M, Moracci M
Year: 2011
A novel α-d-galactosynthase from Thermotoga maritima converts β-d-galactopyranosyl azide to α-galacto-oligosaccharides
Cobucci-Ponzano B, Zorzetti C, Strazzulli A, Carillo S, Bedini E, Corsaro MM, Comfort DA, Kelly RM, Rossi M, Moracci M
Year: 2011
Engineering the stability and the activity of a glycoside hydrolase
Cobucci-Ponzano B, Perugino G, Rossi M, Moracci M
Year: 2010
Recent developments in the synthesis of oligosaccharides by hyperthermophilic glycosidases
Moracci M, Cobucci-Ponzano B, Perugino G, Giordano A, Trincone A, Rossi M
Year: 2005




